Industrial HEPA filters or High-Efficiency Particle Air filters are an essential part of the pharmaceutical and healthcare industries, where sterile environments are mandatory. Particles that cannot be seen by the naked human eye fill the air. These minute particles can cause the development of problems when working with new medicines and medical equipment as well as carrying airborne disease.
The smaller the micron, the harder it is to filter out of the air. To put it into perspective the human eye can detect a particle that is approximately 10 microns. Pollen and plant spores are in this range and hence visible to our eye with some precise detection. Bacteria can be as small as 0.3 microns, which is why 0.3 micron industrial HEPA filters are used in pharma companies.
A significant reason why a 0.3 micron HEPA is used in pharma companies in research labs where the researchers and medical staff are working with bacteria and viruses air needs to be as clean as it can be to contain the research particles. Virus particles are smaller than 0.3 microns, but generally, they travel on larger particles such as saliva or sweat.
Related: Basics of HVAC System
In operating theatres and emergency rooms where open wounds are being handled, it is essential for the wound to be in contact with as little exposure to particles that could result in infection.
Related: Working of HVAC System and Its Functions
How do HEPA filters work?
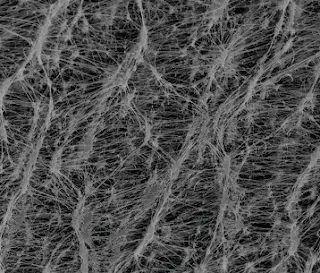
The HEPA system is a 4 step process that starts by filtering out the larger particles with a sieving process as well as inertial impaction. The air then flows through a finer type of mesh fiber that will trap most of the finer particles. To complete the process, the air then moves through the filter and will be caught by colliding with the fiber. When the air comes through the other side of the HEPA, it is supposed to have taken out 99.97% of particles from the air.What does HEPA make It different from other Filter Systems?
A standard air filter usually eliminates medium to larger particles from the air however they do not have the capabilities to almost completely eradicate tiny micron size particles from the air as a HEPA does.The smaller the micron, the harder it is to filter out of the air. To put it into perspective the human eye can detect a particle that is approximately 10 microns. Pollen and plant spores are in this range and hence visible to our eye with some precise detection. Bacteria can be as small as 0.3 microns, which is why 0.3 micron industrial HEPA filters are used in pharma companies.
A significant reason why a 0.3 micron HEPA is used in pharma companies in research labs where the researchers and medical staff are working with bacteria and viruses air needs to be as clean as it can be to contain the research particles. Virus particles are smaller than 0.3 microns, but generally, they travel on larger particles such as saliva or sweat.
Related: Basics of HVAC System
In operating theatres and emergency rooms where open wounds are being handled, it is essential for the wound to be in contact with as little exposure to particles that could result in infection.
Manufacturer Requirements to meet the Pharma Companies Standards
Each and every HEPA has to be individually tested to ensure that there are absolutely no faults in each one. Even the tiniest defect could defeat the purpose of the HEPA. After the manufacturing process is completed, the product is tested and is certified.Related: Working of HVAC System and Its Functions


It's good helpful for freshers to improve knowledge on pharm.
ReplyDeletePl contact me or give me ur contact
ReplyDelete